|
This video shows a sample of cesium-137 being tested for emission of radiation. Nuclei of cesium-137 isotopes are unstable and will undergo a process of radioactive decay to form more stable isotopes. Cesium-137 is formed during fission reactions of uranium-235 in nuclear power plants and nuclear weapons. The equation that describes the radioactive decay of cesium is shown below. The conversion of a neutron (from the cesium nucleus) to a proton causes the atomic number to increase from 55 to 56. This leads to the formation of barium-137. It's important to notice that the mass number stays the same in examples of beta decay.
0 Comments
Leave a Reply. |
Noel PaullerThank for visiting my blog where I post thoughts from the random corner of the universe which is my brain. Archives
October 2022
|


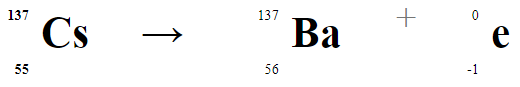
 RSS Feed
RSS Feed
