|
HS-PS2-5: Plan and conduct an investigation to provide evidence that an electric current can produce a magnetic field and that a changing magnetic field can produce an electric current. (HS-PS2-5 Evidence Statements) The NGSS performance expectations are basically asking that students re-create the experiments of Michael Faraday. In 1831, Faraday discovered the relationship between electricity and magnetism. His experiments showed that electric currents could be manipulated to create magnetic fields and magnetic fields could be manipulated to create electric currents. I recently had my students compete in an engineering challenge as they worked towards meeting this learning standard. The students really enjoyed having the opportunity to explore the topics of electricity and magnetism, design and build their electromagnets, test them and then finally compete. Students were allowed to use the following materials. Students were allowed to request additional or modified materials as needed.
The competition consisted of two parts. The first challenge required students to activate their magnet and lift as many regular size paper clips as their magnet could support. The second part of the challenge required that students lift a single paper clip at a time and transfer it a distance of 1 meter and drop it into a shoe box without pulling the paper clip off with their hands or "scraping" it off into the box. Groups had 90 seconds to transfer as many paper clips as possible. Groups were given a score for each challenge based on their overall rank, with the top group in each challenge receiving a score of 1. The grand champion was determined by the group that had the lowest point total.
0 Comments
This video shows a sample of cesium-137 being tested for emission of radiation. Nuclei of cesium-137 isotopes are unstable and will undergo a process of radioactive decay to form more stable isotopes. Cesium-137 is formed during fission reactions of uranium-235 in nuclear power plants and nuclear weapons. The equation that describes the radioactive decay of cesium is shown below. The conversion of a neutron (from the cesium nucleus) to a proton causes the atomic number to increase from 55 to 56. This leads to the formation of barium-137. It's important to notice that the mass number stays the same in examples of beta decay. NGSS HS-PS3-3: Design, build, and refine a device that works within given constraints to covert one form of energy into another form of energy. For a number of years I taught a 9th grade physical science course that included a unit about engineering. The final project of the unit had students designing, building, and testing trebuchets. The students would compete to see which group could launch a projectile the greatest distance. The competition could be ramped up by adding in an accuracy component as well.
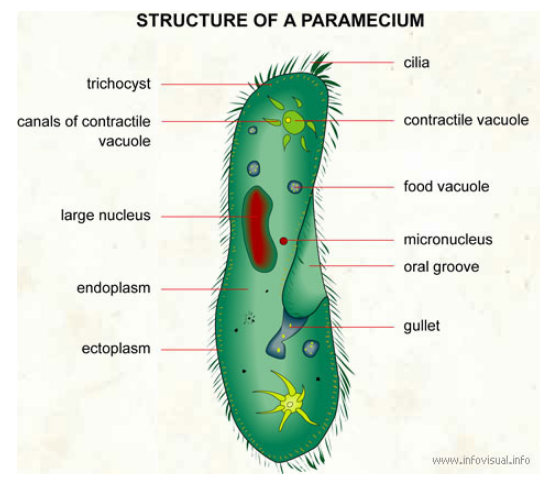
Students learned that the counterweight possesses gravitational potential energy which is converted to kinetic energy when the counterweight falls. The dropping counterweight makes the throwing arm whip around to throw a projectile. This activity takes 2-3 class periods. When I have had to complete this activity in fewer class period I pre-make the throwing arm and we would just switch it from one build to the next during testing. If you have more time I think it makes the activity more challenging for students to need to figure out how to make the throwing arm. There are many options for constraint for this activity. I limited my students to a 1kg counterweight. They were also given set amounts of cardboard, dowel, paper, and had to complete their build within a time limit. The video shown below is from the winning student trebuchet from one of my classes during the 2015-2016 school year at Coon Rapids High School. The materials used for this activity included: -packing tape -cardboard (from cereal boxes) -wooden dowel -paper clips -string -single hole rubber stopper -paper HS-PS1-2: Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the patterns of chemical properties. HS-PS1-2 Evidence Statements The video below shows the example of magnesium reacting with oxygen in a combustion reaction. When magnesium and oxygen react they form an ionic compound. This is due to the fact that magnesium is a metallic element and oxygen is nonmetallic. As an alkaline earth metal (group II) on the periodic table magnesium has two valence electrons it will donate when forming a 2+ cation. Oxygen (group VI) has six valence electrons and will gain two electrons when forming a 2- oxide anion. The magnesium and oxide ions will therefore combine in a 1:1 ratio. The balanced equation for this reaction is shown below. Answer: Paramecium can be found in pond water. Explanation:Paramecium can also be found in brackish and salt water. These protists are single celled organisms. Here is a picture of a paramecium. The video below shows several paramecium feeding. They are using their cilia, which are found all along the outside of the cell membrane, for movement. I hope this helps! HS-LS4-3: Apply concepts of statistics and probability to support explanations that organisms with an advantageous heritable trait tend to increase in proportion to organisms lacking this trait. HHMI BioInteractive video about the Rock Pocket mouse of the American southwest is a great example of how natural selection can cause evolutionary change.
Follow the link below for supporting documents for a classroom activity to support student learning of this topic. Developing an Explanation for Mouse Fur Color Another activity I've used to help students learn this concept is the Evodot simulation created by Jon Herron. Evodot Materials One of the really important HS-LS2-5 performance expectations is that students know the inputs and outputs of cellular respiration. The video below shows a lab I've used for years with my biology students to teach this concept. The word equation for cellular respiration is summarized below. glucose (sugar) + oxygen -> carbon dioxide + water Students perform the experiment using yeast and apple juice. I have also used sugar water or solutions of different sugars. The three trials the students typically do include... 1. warm water 2. room temperature water 3. cold water The lab also provides an opportunity to review or introduce factors can affect the speed of a chemical reaction (HS-PS1-5)
This video shows a classic lab examining the effect of concentration of reactants on the rate of a chemical reaction. This video supports NGSS HS-PS1-5.
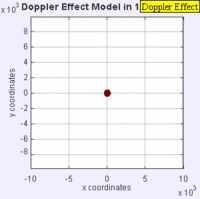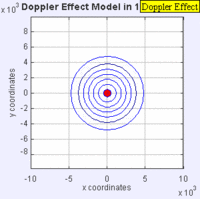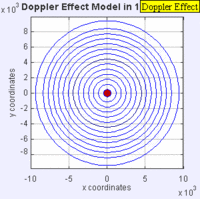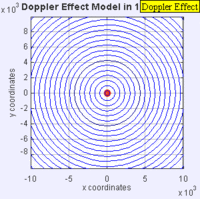
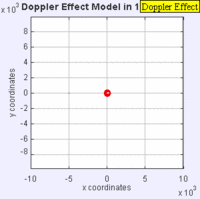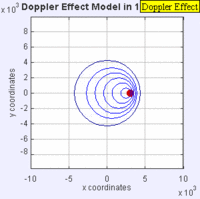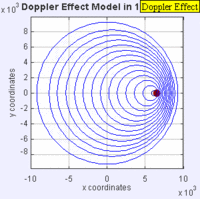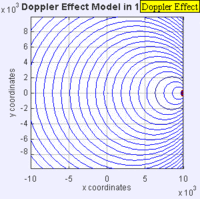
In this experiment solutions of sodium thiosulfate and hydrochloric acid are combined in a beaker. The reaction produces a precipitate which eventually makes the liquid turn cloudy. The reaction beaker is placed over a piece of paper with a letter "X" printed on it. The reaction is said to have gone to completion when the "X" can no longer be seen through the liquid in the beaker. I have used this video as a warm up or as a flipped lesson which has students make observations, record data about the reaction time for the trials, create a graph and form a conclusion about the effect concentration has on the speed of a chemical reaction. This lab was originally published by Flinn Scientific as a Chem Fax! Use this link to see the full description of the experiment. To understand the evidence that the universe is getting larger, we need to understand the Doppler effect. This is the reason why a siren from a police car or fire truck has a higher pitch as it is approaching and a lower pitch when it is traveling away from us. The two images below are really useful in trying to better understand the Doppler effect. The first image has a stationary red dot which is giving off waves, which is shown by the concentric blue circles. The wavelengths of the waves being given off are represented by the distance between the concentric blue circles. The waves would appear the same to four observers each located at the corners of the square area. The second image shows waves being produced by a moving object. In this case, the waves would appear to be different to observers on the right or left side of the square area. Observers on the left side would observe waves that are stretched out because the object giving off the waves is moving away from them. Longer wavelength sounds have lower pitches. An observer on the right side of the square would observe waves that are compressed. Shorter wavelength sounds have higher pitches. So the siren of a police car which is driving towards you will be heard as a higher pitch while the siren of the same car moving away from you will be heard as a lower pitch! By Aleš Tošovský (Own work) [GFDL, CC-BY-SA-3.0 or CC BY-SA 2.5-2.0-1.0], via Wikimedia Commons The Doppler effect also impacts the wavelengths of light being emitted from stars. The light we see from stars that are moving away from us has wavelengths which are stretched out. This does not mean that all the light from this star appears red, just that the spectral lines from this star (almost like a DNA fingerprint for a star) are shifted in the direction of red light. Notice how the black vertical lines have moved to the right in the middle (Redshifted) spectra as compared with the top (unshifted) spectra. Science has concluded that distant galaxies are moving farther and farther away from us because the light from those galaxies is redshifted. By extension science has used this data to conclude that all of the known universe is getting bigger because of continued expansion that started with the Big Bang.
I hope this was helpful! HS-PS2-6: Communicate scientific and technical information about why the molecular-level structure is important in the functioning of designed materials. PS2-6 Evidence Statements I introduced this topic to students by showing them a short video about gecko adhesive (synthetic setae). Geckos feet are super-sticky due to numerous Van der Waals forces that form between the tiny endings of hairs that are found under their feet and whatever surface they are climbing on. After the video we discussed the importance of molecular level interactions for the designed materials shown in the video. Video options include the Weird Connections Episode "How Sticky is Your Gecko?", the segment about gecko adhesive from Nova's "Making Stuff Smarter" or "Bio-Inspiration:Nature as Muse" from KQED-Quest. By Bjørn Christian Tørrissen - Own work by uploader, http://bjornfree.com/galleries.html, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=6874833
Brainstorm list of materials students could research to meet this standard.
|
Noel PaullerThank for visiting my blog where I post thoughts from the random corner of the universe which is my brain. Archives
October 2022
|
||||||||



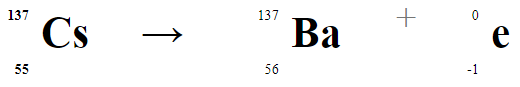
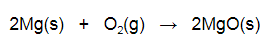


 RSS Feed
RSS Feed
