|
HS-PS1-2: Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the patterns of chemical properties. HS-PS1-2 Evidence Statements The video below shows the example of magnesium reacting with oxygen in a combustion reaction. When magnesium and oxygen react they form an ionic compound. This is due to the fact that magnesium is a metallic element and oxygen is nonmetallic. As an alkaline earth metal (group II) on the periodic table magnesium has two valence electrons it will donate when forming a 2+ cation. Oxygen (group VI) has six valence electrons and will gain two electrons when forming a 2- oxide anion. The magnesium and oxide ions will therefore combine in a 1:1 ratio. The balanced equation for this reaction is shown below.
0 Comments
Leave a Reply. |
Noel PaullerThank for visiting my blog where I post thoughts from the random corner of the universe which is my brain. Archives
October 2022
|


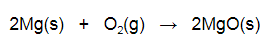
 RSS Feed
RSS Feed
